Online first articles
Articles not assigned to any issue
CLINICAL CASE REPORT
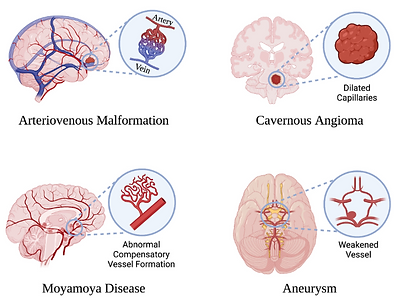
Acute right brainstem infarction complicated by decompensated liver cirrhosis, coagulopathy, and progressive thrombocytopenia
Lang Jiayu,Zhao Miaomiao,Zheng Xin,Zhang Sumei
Acute cerebral infarction is a common neurological emergency associated with high morbidity and mortality. Management becomes particularly challenging in patients with decompensated liver cirrhosis, who frequently present with coagulopathy and thrombocytopenia, thereby carrying simultaneous risks of hemorrhage and thrombosis. These competing clinical risks substantially complicate antiplatelet treatment decisions. We report a case of acute right brainstem infarction in a patient with hepatitis B–related decompensated liver cirrhosis, prior transjugular intrahepatic portosystemic shunt (TIPS) placement, coagulopathy, and progressive thrombocytopenia. Owing to the patient’s markedly elevated bleeding risk, antiplatelet therapy was initially withheld. Conservative management was undertaken, including plasma transfusion to improve coagulation status, acid suppression and gastric mucosal protection, and maintenance of cerebral perfusion, accompanied by close dynamic monitoring of coagulation parameters and platelet counts. This case highlights the complexities of individualized clinical decision-making in patients with acute cerebral infarction complicated by advanced liver disease and severe hematological abnormalities. It further underscores the importance of balancing thrombotic and hemorrhagic risks in developing safe and effective management strategies for this highly vulnerable patient population.

PERSPECTIVE
From bioinformatics to translational neuroscience: computational modeling at the frontier of drug discovery
Filippo Dall'Armellina
The prediction of three-dimensional protein structures has undergone a paradigm shift, driven primarily by deep learning-based tools. These advances are beginning to permeate neuroscience research, offering new routes for understanding the molecular basis of neurological disease and accelerating early-stage central nervous system (CNS) drug discovery. This article examines the current state of in silico structural modeling as it applies to translational neuroscience, highlighting areas of progress — including G protein-coupled receptor (GPCR) pharmacology, cryo-EM-informed structural neurobiology, protein aggregation in neurodegeneration, and AI-driven small molecule discovery — alongside discussion of limitations. These include the misinterpretation of static computational models, and the continuing gap between structural insight and clinical validation. The argument advanced here is that structural modeling has already meaningfully altered the landscape of early drug discovery, but that its translational promise will only be realised through sustained interdisciplinary integration and rigorous experimental follow-through.
ORIGINAL RESEARCH
Mutations of TARDBP gene: conformational changes of TDP43 protein occurring in Amyotrophic Lateral Sclerosis disease
Martina Cerrato, Laura D’Aiello, Alessandra Darino, Elisa Maria Gatti1, Fabio Persia, Jack A. Tuszynski
Amyotrophic Lateral Sclerosis (ALS) is strongly associated with TDP43 proteinopathy, where mutations in TARDBP gene alter the protein’s stability, solubility, and RNA related functions. This study examines eight ALS-linked missense mutations located in both the structured RRM domains and the intrinsically disordered C-terminal region, aiming to clarify how single residue substitutions perturb TDP43 structure and behavior. The full length TDP43 model was obtained from the AlphaFold database due to the absence of complete crystallographic structures. Each mutation was manually introduced into the appropriate domain using Molecular Operating Environment (MOE) software. Under appropriate conditions, Molecular Dynamics simulations were performed on wildtype and mutated structures; Wildtype and mutant proteins were then compared with respect to conformational changes, focusing on energetic profiles, surfaces and electrostatics properties, considering how these variations can be related to effects of mutations found in literature. RRM domain mutations showed localized but mechanistically distinct alterations. D169G increased domain stability through a subtle β-turn rearrangement without affecting nucleic acid binding. K181E and K263E reversed local electrostatics, disrupting the positively charged RNA binding groove. C terminal mutations (Q331K, A315E, M337V, N345K, G298S) produced broader effects, including enhanced aggregation propensity, altered phase behavior, impaired DNA repair interactions ...

ORIGINAL RESEARCH
Focusing on a novel upstream target for Alzheimer’s Disease therapy: Identification of potential BACE1 inhibitors Using In Silico Methods
Matteo Bulgini and Jack A. Tuszynski
Alzheimer’s Disease (AD) is the most common form of dementia and one of the most prevalent neurodegenerative diseases in the world. Currently, there’s no definitive cure for this disease. The most widely accepted hypothesis involves the accumulation of β-amyloid into plaques and of the MAP τ protein into neurofibrillary tangles located between and inside the neurons, respectively. These processes cause neuronal degeneration and a disruption of the functionality of the brain. The β-amyloid plaques are due to the action of the β -secretase protein, which cleaves the APP protein in the Aβ region, resulting in β -amyloid fragments that aggregate, as opposed to the production of α-amyloid, which can stimulate neuroplasticity and neuroregenerative capabilities. Previous research showed that mutations in the sequence of the APP protein can inhibit the formation of these fragments, but this route is not conducive to therapeutic approaches. Several trials have been conducted into developing pharmaceutical compounds that can inhibit the cleavage site and in particular the catalytic dyad (Asp32−Asp228). Using the data from these trials, the aim of the research reported in this paper was to widen the possibilities of drug discovery by parsing a wide database of bioactive molecules, simulating their interactions with the BACE1 protein. By forming bonds with the pocket responsible for the cleavage of the APP protein, these ligands may be putative inhibitors. Here, we report the identification of several bioactive compounds predicted to be active inhibitors of the β-secretase 1, which is the main enzyme responsible for the formation of the β amyloid plaques in the pathogenesis of AD. To this end we performed database parsing, selecting clinical-stage drugs and similar compounds, which then were computationally docked to a mature chain of BACE1 protein using Boltz2 AI-based software. From the resulting compounds the top 27, ranked for confidence, affinity and accounting for the wider base possible, were then investigate for binding affinity to the target using the GROMACS suite to predict their docking poses to the protein. This was done by calculating free energy applying the MM-GBSA algorithm for the resulting structures. The highest ranked compounds can be validated by in vitro and in vivo studies and potentially represent drug candidates for AD therapies.

ORIGINAL RESEARCH
Intracellular Calcium Dynamics in Starburst Amacrine Cell Dendrites:
The Onset of Cardinal Direction Selectivity and Speed Tuning*
N.L. Iannella
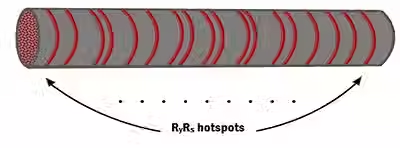
Detecting moving objects is crucial in the animal kingdom and is fundamental to vision. In the vertebrate retina, starburst amacrine cells are directionally selective in terms of their calcium responses to stimuli that move centrifugally from the soma. The mechanism by which starburst amacrine cells show calcium bias for centrifugal motion is still to be determined. Recent morphological studies using fluorescent microscopy and immunostaining have shown that the endoplasmic reticulum is omnipresent in the soma, extending to the distal processes of starburst amacrine cells. Electron microscopy for ChAT SAC in adult rat retina unequivocally proves the presence of local endoplasmic reticulum. The submicron in diameter dendrites implies that the endoplasmic reticulum is not luminally connected between the soma and the distal tips. We construct a computational model of SAC dendrites with ER to simulate the Ca2+-induced Ca2+ release (CICR)-based calcium waves in the presence of unsaturated buffer to test the hypothesis that CICR mechanism can sustain constant calcium wave propagation in the centrifugal direction. The veto mechanism with a 100msec delay for the operation of retinal direction selectivity. is a working hypothesis, in which a CICR mechanism in the presence of local endoplasmic reticulum underlies speed tuning for directionality and propagation failure in the centripetal direction due to a build-up of calcium hyperexcitability in the distal regions of starburst amacrine cells. Modeling the heterogeneity of calcium endoplasmic reticulum in simulated starburst amacrine cells sheds light on a possible explanation for the cause ...
.png)